The Severe Mental Illness Longitudinal Evaluation (SMILE) BioResource is a re-contactable resource of volunteers with severe mental illnesses.

A collaboration led by the Mental Health Translational Research Collaboration Mission, the University of Oxford (Oxford Health Biomedical Research Centre) and the NIHR BioResource to investigate the causes of severe mental illness (SMI) and how best to develop new treatments.
The SMILE BioResource launched in November 2025 and will collect information from people with experience of SMI to provide a resource for researchers to investigate the risk of SMI, its symptoms and how to manage difficult side effects of medication that some people experience.
Severe mental illness is a term that includes mental health disorders such as:
People with SMI may experience short-term and long-term issues, and current medication and other treatments may not work well enough.
A longitudinal study means that participants are included in a study over a period of time, and that additional data may be collected from them. A benefit of longitudinal investigation is that researchers may be able to discover developments or changes in characteristics over time.
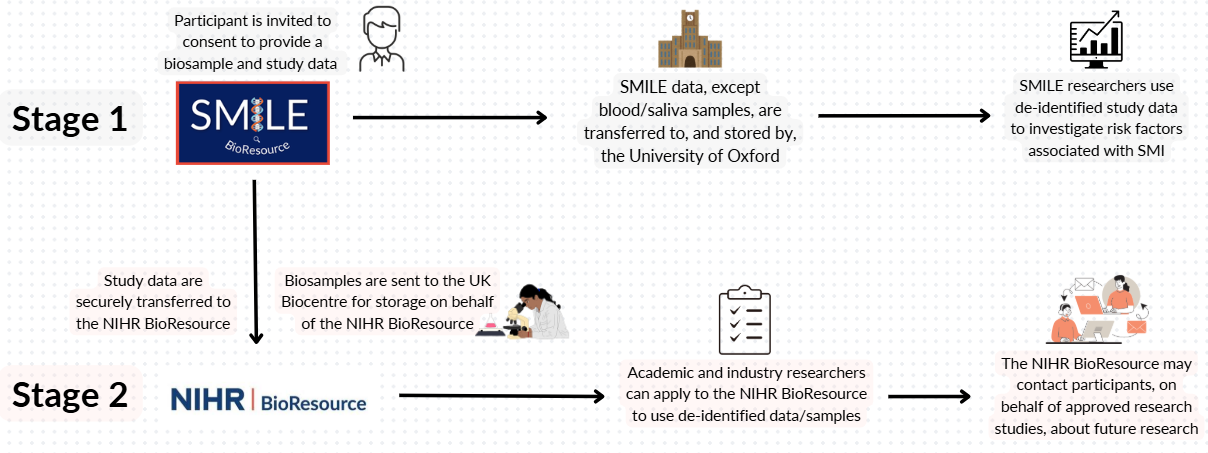
The SMILE BioResource is part of the NIHR BioResource, which includes hundreds of thousands of volunteers who wish to participate in health research, with and without health conditions. Volunteers joining the BioResource are asked to donate a small blood or saliva sample and to give consent to be invited to participate in health-related research based on data they have provided.
Yes, participants may bring a close relative or friend, care coordinator or peer support worker to the study visit for additional support.
No, unless a participant discloses that they intend to harm themselves. It is the duty of the personnel leading the study visit to pass this information on to a care provider (such as a secondary mental health team and/or GP) so that appropriate care can be put in place. Otherwise, participation in this study is completely separate from the care that you receive from your GP.
All information collected will be handled according to strict ethical and legal standards to ensure safety and security, including but not limited to:
Find out more information about our safety and security best practices policies and how we keep your information safe.
Researchers aim to study how genes, made of DNA, influence health and disease by analysing DNA and other components from blood or saliva samples. All samples will be securely processed and stored, coded instead of named, though DNA can never be fully anonymous.
When released for a Stage 2 study (after participant consent), samples will be handled confidentially in a non-identifiable form, meaning they cannot be withdrawn afterward. Samples may be stored for up to ten years to allow approved research access and participant invitations to ethically approved future studies.
The University of Oxford processes your information for research under the legal basis of serving the public interest. As the study sponsor, it is responsible for safeguarding and properly using your data. Information may come from medical, hospital, educational, social care, and disease registry records, and will be shared only with the NIHR BioResource and approved researchers. Identifiable details will be replaced with coded numbers, and all data will be stored securely with access limited to authorised personnel. Your information will be retained for up to ten years after the NIHR BioResource closes to support ongoing research objectives.
Your data may be shared outside the UK for health-related studies approved by the NIHR BioResource, but only the necessary information will be released. Efforts will be made to ensure you cannot be identified, though rare conditions may still pose a risk of identification. Approved researchers from public, charitable, commercial or pharmaceutical organisations may access the data under strict conditions.
Protections include:
We also have strict procedures in place to handle suspected data breaches, with notifications provided to you and regulators when legally required.
You can withdraw from the study and request that your data are removed at any point. If you choose to withdraw, you have two options:
Your personal information will be retained in an archive so that a record remains of your initial consent and the withdrawal process. If the SMILE BioResource/ NIHR BioResource is unable to confirm your decision, your sample(s) and data will be retained for future use, and you will not be contacted again.
You can withdraw from the study at any time.
For further information about this study, please email the study project coordinator (smibioresource@psych.ox.ac.uk) or the Chief Investigator (rachel.upthegrove@psych.ox.ac.uk).
You can also contact the NIHR BioResource team with any queries on 0800 090 2233 or email: nbr@bioresource.nihr.ac.uk.